Chemical amplification
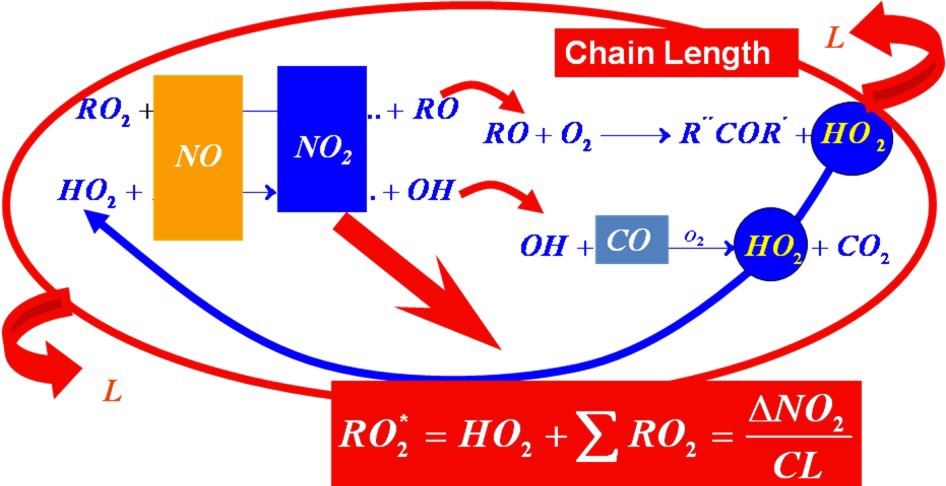
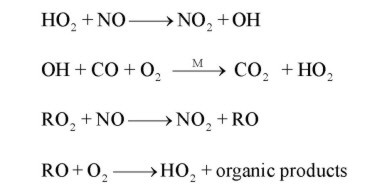
The Peroxy Radical Chemical Amplification (PeRCA) is the most widely technique used for in situ ground based measurements of peroxy radicals. Briefly, PeRCA utilises the amplified conversion of oxy, alkoxy, hydroxyl and alkyl peroxy radicals into the more stable NO2, via a chain reaction involving NO and CO.
|
||
The NO2 is generally measured by detecting the chemiluminiscence of its reaction with luminol (3-aminophthalhydrazide: C8H7N3O2) on a fiber filter paper at the detector. Chemical interferences associated with luminol detectors are minimised by determining the peroxy radical concentrations on the basis of a modulated signal. Thus, the chain chemistry is switched on and off by adding CO at the front (amplification mode) and at the back (background mode) of the reactor, and the total radical concentration is calculated from the difference between the amplification and background signals, DNO2, provided that the amplification factor or chain length (CL) in the reactor is known. As OH and RO abundances in the troposphere are much lower than the rest, PerCA measures to a good approximation the total sum of peroxy radicals (RO2*= HO2+ SRO2). The CL must be determined for each particular set up by using calibration procedures adapted to the measuring purposes. The extent of the chain reaction results from the competition between amplification, chemical loss reactions and wall loses and therefore depends on:
- concentration of the reactants added for the chemical conversion, i.e., NO and CO
- reactor volume, which defines the sample residence time
- reactor material and shape, affecting heterogeneous radical losses
- relative humidity. This newly discovered effect has a significant impact in the determination of RO2* mixing ratios in the boundary layer and might be especially critical for the assessment of nocturnal radical levels. TROLAS has actively participated in the characterisation of the RH dependence of the CL.
The overall accuracy of PeRCA is constrained by the signal stability of the luminol detector. Main instabilities derived from the luminol flow, temperature and pressure variabilities. In an attempt to overcome shortcomings of luminol detectors, TROLAS presently works on the use of cavity enhanced spectroscopy for the detection of NO2.